NCERT Solutions for Class 12 Chemistry Chapter 10 Biomolecules updated for CBSE and State board session 2026-27 in Hindi and English Medium. Class 12 Chemistry unit 10 solutions and concept videos are very helpful for all the students studying chemistry in intermediate.
Viva Question for Class 12 Chemistry
NCERT Solutions for Class 12 Chemistry Chapter 10
Class 12 Chemistry Chapter 10 Biomolecules Question Answers
| Class: 12 | Chemsitry |
| Chapter: 10 | Biomolecules |
| Study Material: | Intext and Exercises Solutions |
| Content Format: | PDF, Text, Images and Videos |
| Session: | Academic Year 2026-27 |
| Medium used: | Hindi and English |
Biomolecules
A biomolecule is any natural particle that is created by a living organism, including enormous polymeric molecules, for example, proteins, polysaccharides, and nucleic acids just as small molecules, for example, primary metabolites, secondary metabolites, and natural products.
Carbohydrates
Carbohydrates are primarily produced by plants and form a very large group of naturally occurring organic compounds. Some common examples of carbohydrates are cane sugar, glucose, starch, etc. Most of them have a general formula, Cₓ(H₂O)ᵧ, and were considered as hydrates of carbon from where the name carbohydrate was derived. For example, the molecular formula of glucose (C₆H₁₂O₆) fits into this general formula, C₆(H₂O)₆.
Class 12 Chemistry Chapter 10 MCQ
Which of the following polymer is stored in the liver of animals?
The functional group which is found in amino acids are
Which of the following compounds is found abundantly in nature?
Proteins are found to have two different types of secondary structures viz. α-helix and β-pleated sheet structure. α-helix structure of protein is stabilised by
Classification of Carbohydrates
Carbohydrates are classified on the basis of their behaviour on hydrolysis. They have been broadly divided into following three groups.
- Monosaccharides: A carbohydrate that cannot be hydrolysed further to give simpler unit of polyhydroxy aldehyde or ketone is called a monosaccharide. About 20 monosaccharides are known to occur in nature. Some common examples are glucose, fructose, ribose, etc.
- Oligosaccharides: Carbohydrates that yield two to ten monosaccharide units, on hydrolysis, are called oligosaccharides. They are further classified as disaccharides, trisaccharides, tetrasaccharides, etc., depending upon the number of monosaccharides, they provide on hydrolysis.
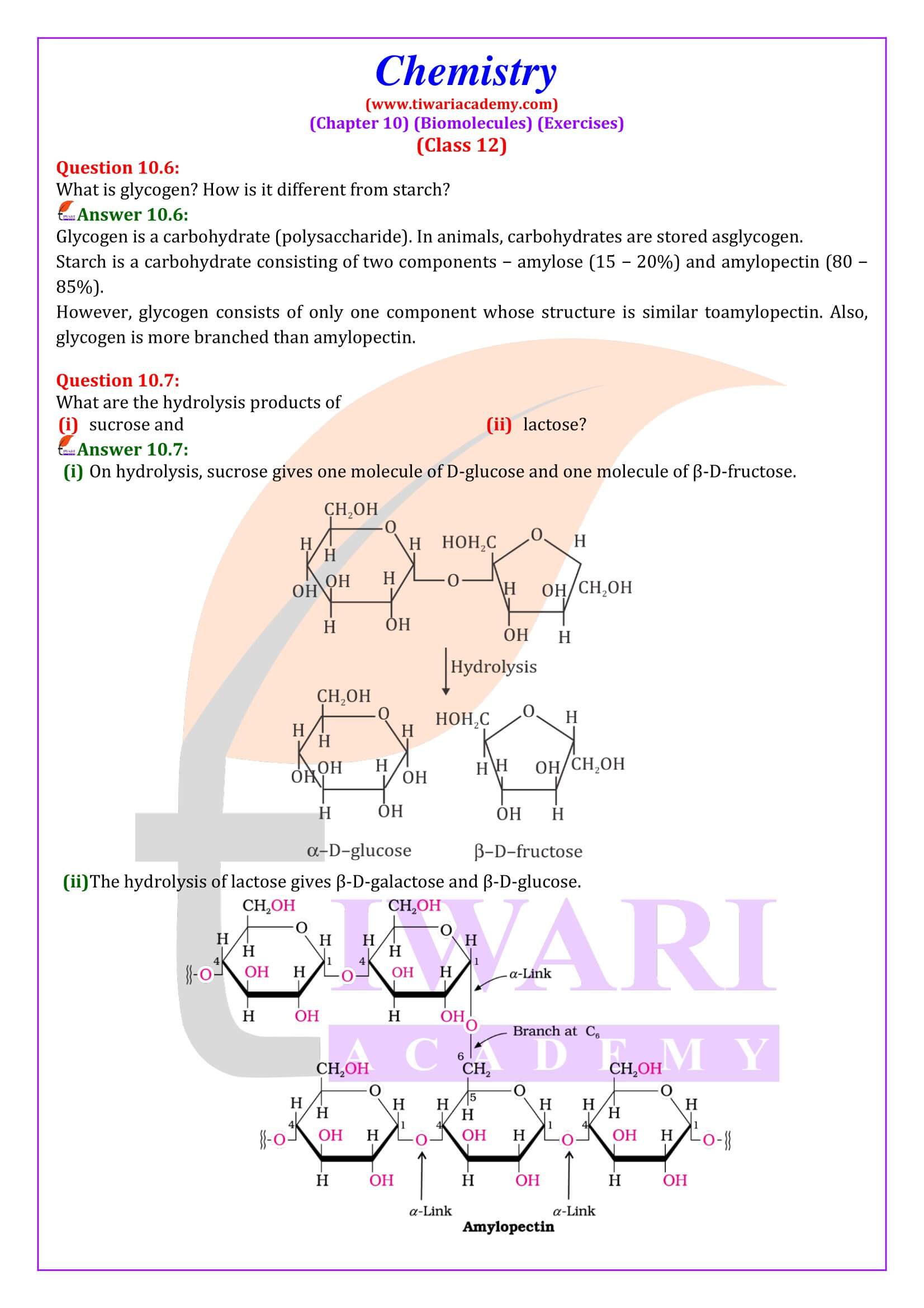
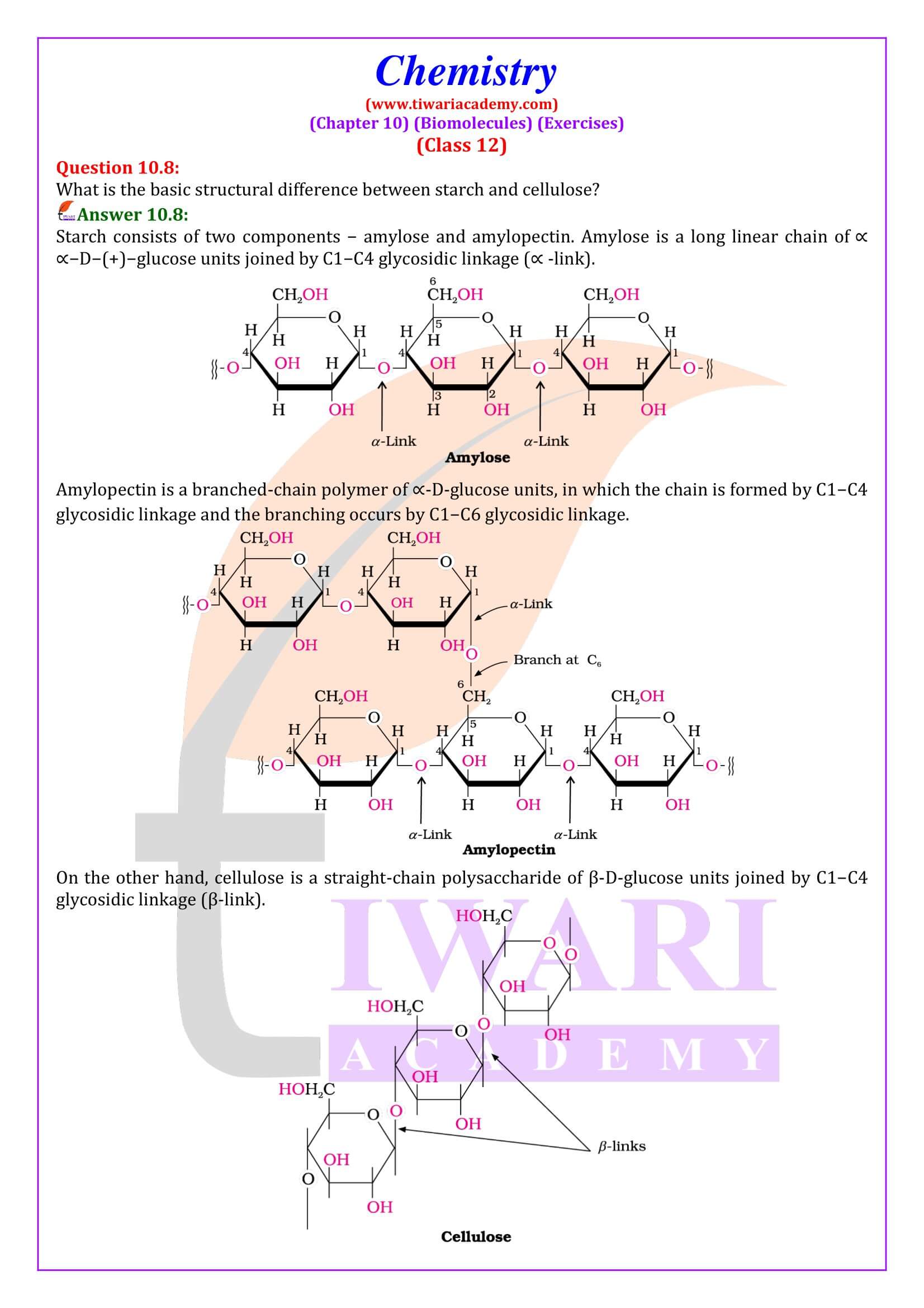
- Polysaccharides: Carbohydrates which yield a large number of monosaccharide units on hydrolysis are called polysaccharides. Some common examples are starch, cellulose, glycogen, gums, etc. Polysaccharides are not sweet in taste; hence they are also called non-sugars.
12th Chemistry Unit 10 Multiple Choice Questions
Starch is composed of two polysaccharides which are
The vitamins absorbed from intestine along with fats are
Carbohydrates are stored in human body as the polysaccharide
Which of the following acids is a vitamin?
Importance of Carbohydrates
Carbohydrates are essential for life in both plants and animals. They form a major portion of our food. Carbohydrates are used as storage molecules as starch in plants and glycogen in animals. Cell wall of bacteria and plants is made up of cellulose. We build furniture, etc. from cellulose in the form of wood and clothe ourselves with cellulose in the form of cotton fibre. They provide raw materials for many important industries like textiles, paper, lacquers and breweries.
Proteins
Proteins are the most abundant biomolecules of the living system. Chief sources of proteins are milk, cheese, pulses, peanuts, fish, meat, etc. They occur in every part of the body and form the fundamental basis of structure and functions of life. They are also required for growth and maintenance of body.
Amino Acids
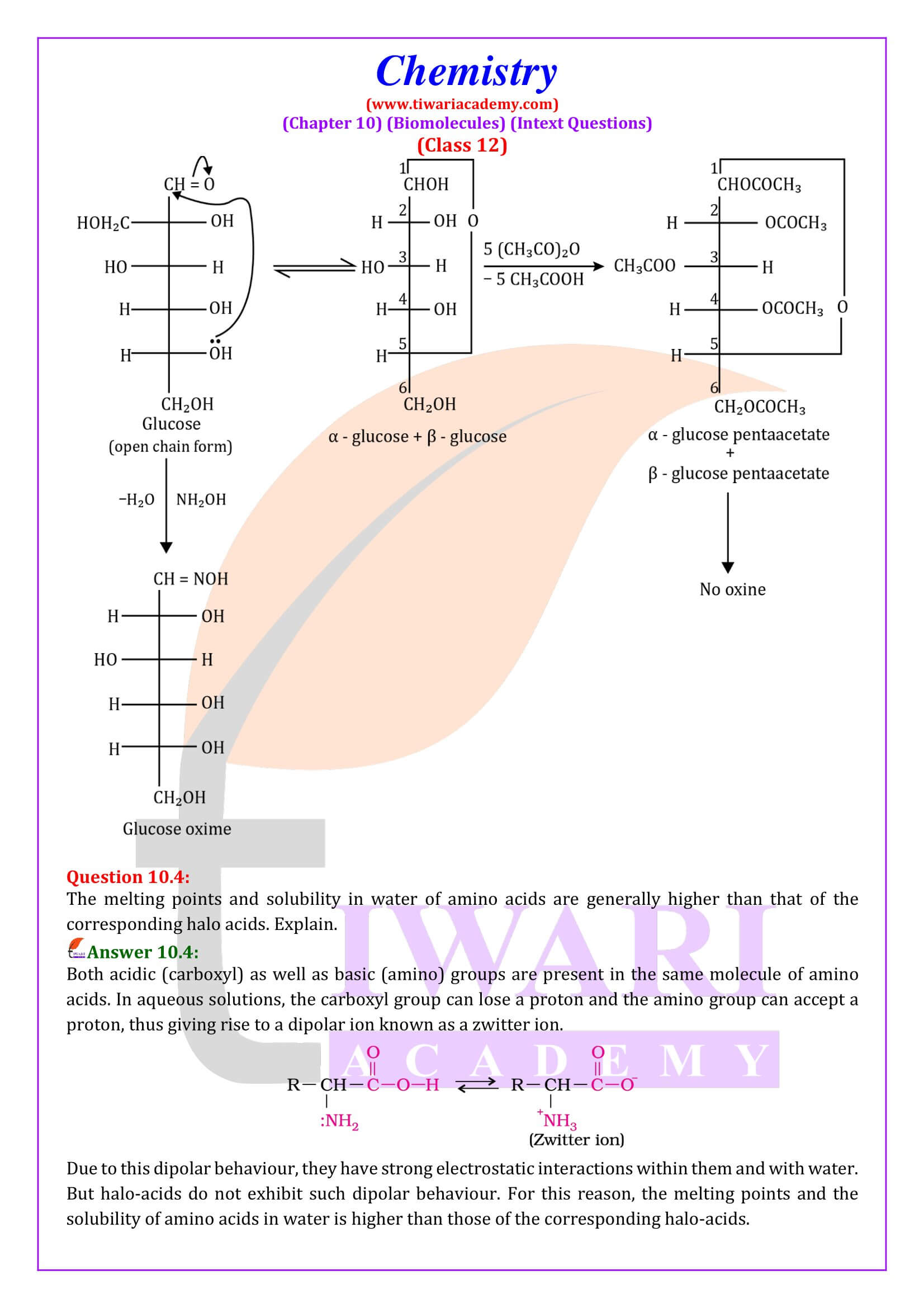
Amino acids contain amino (–NH₂) and carboxyl (–COOH) functional groups. Depending upon the relative position of amino group with respect to carboxyl group, the amino acids can be classified as a, b, g, d and so on. Only a-amino acids are obtained on hydrolysis of proteins. They may contain other functional groups also.
Enzymes and Vitamins
Enzymes
Enzymes are proteins that help speed up chemical reactions in our bodies. Enzymes are essential for digestion, liver function and much more. Too much or too little of a certain enzyme can cause health problems. Enzymes in our blood can also help healthcare providers check for injuries and diseases.
Vitamins
It has been observed that certain organic compounds are required in small amounts in our diet but their deficiency causes specific diseases. These compounds are called vitamins. They are generally regarded as organic compounds required in the diet in small amounts to perform specific biological functions for normal maintenance of optimum growth and health of the organism. Vitamins are designated by alphabets A, B, C, D, etc. Some of them are further named as sub-groups e.g. B₁, B₂, B₆, B₁₂, etc.
Class 12 Chemistry Chapter 10 Important Questions
What do you mean by biomolecules?
Definition: A biomolecule is a chemical compound found in living organisms. These include chemicals that are composed of mainly carbon, hydrogen, oxygen, nitrogen, sulfur and phosphorus. Biomolecules are the building blocks of life and perform important functions in living organisms.
What are biomolecules proteins?
Proteins are biomolecules comprised of amino acid residues joined together by peptide bonds. Biomolecules are molecules produced by living organisms. As such, most of them are organic molecules. Proteins are one of the major biomolecules.
What are carbohydrates in biomolecules?
Carbohydrates are the most abundant among the major classes of biomolecules. Carbohydrate (biology definition): any of the group of organic compounds consisting of carbon, hydrogen, and oxygen, usually in the ratio of 1:2:1, hence the general formula: Cₙ(H₂O)ₙ. Synonyms: saccharide, carb.