NCERT Class 9 Science Exploration Solutions Chapter 5 Exploring Mixtures and their Separation – Exercise Question answers for Session 2026-27 updated. Chapter 5 of the Class 9 Science Exploration textbook (NCERT, Session 2026–27), titled Exploring Mixtures and their Separation, introduces students to the science behind classifying and separating mixtures. Starting with how sugar crystals are obtained from sugarcane and how malaria is detected from blood, the chapter covers homogeneous and heterogeneous mixtures, three concentration formulas (% m/m, % m/v, % v/v) and solubility curves.
Separation techniques covered include crystallization, distillation, paper chromatography, separating funnel, sublimation, centrifugation and coagulation. The chapter also explains solutions, suspensions and colloids through particle size and the Tyndall effect. India’s scientific contributions — including Kannauj’s traditional Deg-Bhapka distillation method and the electricity-free paperfuge for malaria detection — make this chapter both culturally rich and practically relevant for Session 2026–27.
Quick Links:
NCERT Class 9 Science Exploration Chapter 5 Solutions
Class 9 Science Exploration Chapter 5 Question Answer
Page 90 – Revise, Reflect Refine
1. Which of the following mixtures are correctly classified as homogeneous (Hm) and heterogeneous (Ht)? Choose the correct option.
(i) Air — Hm, Milk — Ht, Sugar solution — Hm, Smoke — Hm
(ii) Brass — Ht, Fog — Ht, Vinegar — Ht, Muddy water — Hm
(iii) Copper sulphate solution — Hm, Salt solution — Hm, Milk — Hm, Bronze — Hm
(iv) Muddy water — Ht, Milk — Ht, Blood — Ht, Brass — Hm
Answer:
(iv) Muddy water — Ht, Milk — Ht, Blood — Ht, Brass — Hm
Explanation:
- Muddy water → Heterogeneous (particles visible)
- Milk → Colloid (treated as heterogeneous)
- Blood → Heterogeneous (cells + plasma)
- Brass → Homogeneous (alloy).
2. Choose the correct options and explain the reason for the correct and incorrect options.
Which among the following mixtures show the Tyndall Effect?
A mixture of:
(a) Air and dust particles
(b) Copper sulphate and water
(c) Starch and water
(d) Acetone and water
(i) a and b
(ii) b and d
(iii) a and c
(iv) c and d
Answer:
(iii) a and c
Explanation:
Tyndall effect is shown by colloids.
- (a) Air + dust → colloid → shows Tyndall effect
- (b) Copper sulphate solution → true solution → no Tyndall
- (c) Starch + water → colloid → shows Tyndall effect
- (d) Acetone + water → solution → no Tyndall
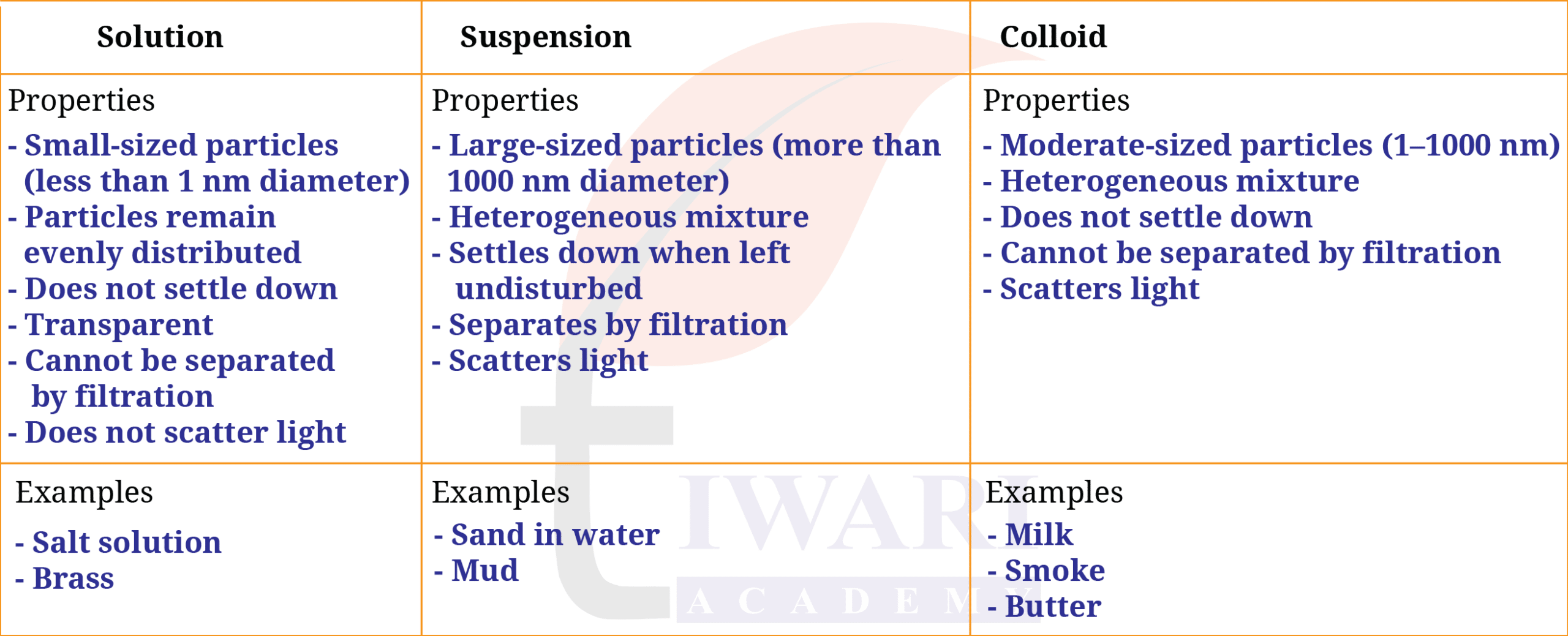
3. A mixture can be categorised as a solution, a suspension, or a colloid, each possessing distinct properties. Utilise the words or phrases provided in the box to fill in the Table 5.2. Words and phrases may be used more than once.
Large-sized particles; Particles remain evenly distributed; Small-sized particles (less than 1 nm diameter); Moderate-sized particles (1 – 1000 nm); Settles down when left undisturbed (more than 1000 nm in diameter); Does not settle down; Scatters light; Separates by filtration; Transparent; Salt solution; Milk; Sand in water; Smoke; Heterogeneous mixture; Cannot be separated by filtration; Mud; Butter; Brass.
image box and Table
Complete the Table 5.2.
Answer:
4. Solve the following problems:
(i) A cake recipe uses dry ingredients, namely 75 g of sugar for 420 g of all-purpose flour and 5 g of sodium hydrogencarbonate. Express the concentration of each component in the mixture using an appropriate method.
Answer:
Total mass = 75 + 420 + 5 = 500 g
Mass percentage:
- Sugar = (75/500) × 100 = 15%
- Flour = (420/500) × 100 = 84%
- Sodium hydrogencarbonate = (5/500) × 100 = 1%
Sugar = 15%, Flour = 84%, Sodium hydrogencarbonate = 1%
(ii) A brass alloy contains 70% copper by mass. Calculate the quantities of copper and zinc present in 120 g of brass.
Answer:
Copper = 70% of 120 = (70/100) × 120 = 84 g
Zinc = 120 − 84 = 36 g
Copper = 84 g, Zinc = 36 g
5. The label on a cooking oil pack says one litre (910 g). If this oil is mixed with water, will it form a separate layer? If so, which substance will be on top? How will you separate the two layers? Also, draw the diagram of the apparatus used.
Answer:
- Yes, oil and water form separate layers because they are immiscible.
- Oil will be on top because it is less dense than water.
- Method of separation: Separating funnel
Reason:
Liquids with different densities that do not mix can be separated using a separating funnel.

6. Assertion (A): Solutions do not exhibit the Tyndall effect. Reason (R): The particles in solutions are larger than 100 nm, so they cannot scatter light.
Choose the correct option:
(i) Both A and R are true, and R is the correct explanation of A.
(ii) Both A and R are true, but R is not the correct explanation of A.
(iii) A is true, but R is false.
(iv) A is false, but R is true.
Answer:
(iii) A is true, but R is false.
Explanation:
- Assertion is true: Solutions do not show Tyndall effect.
- Reason is false: Particles in solutions are very small (< 1 nm), not larger than 100 nm.
7. How would you separate the mixtures given in Table 5.3? Mention the reason for choosing your method. If a mixture cannot be separated, explain why.
Answer:
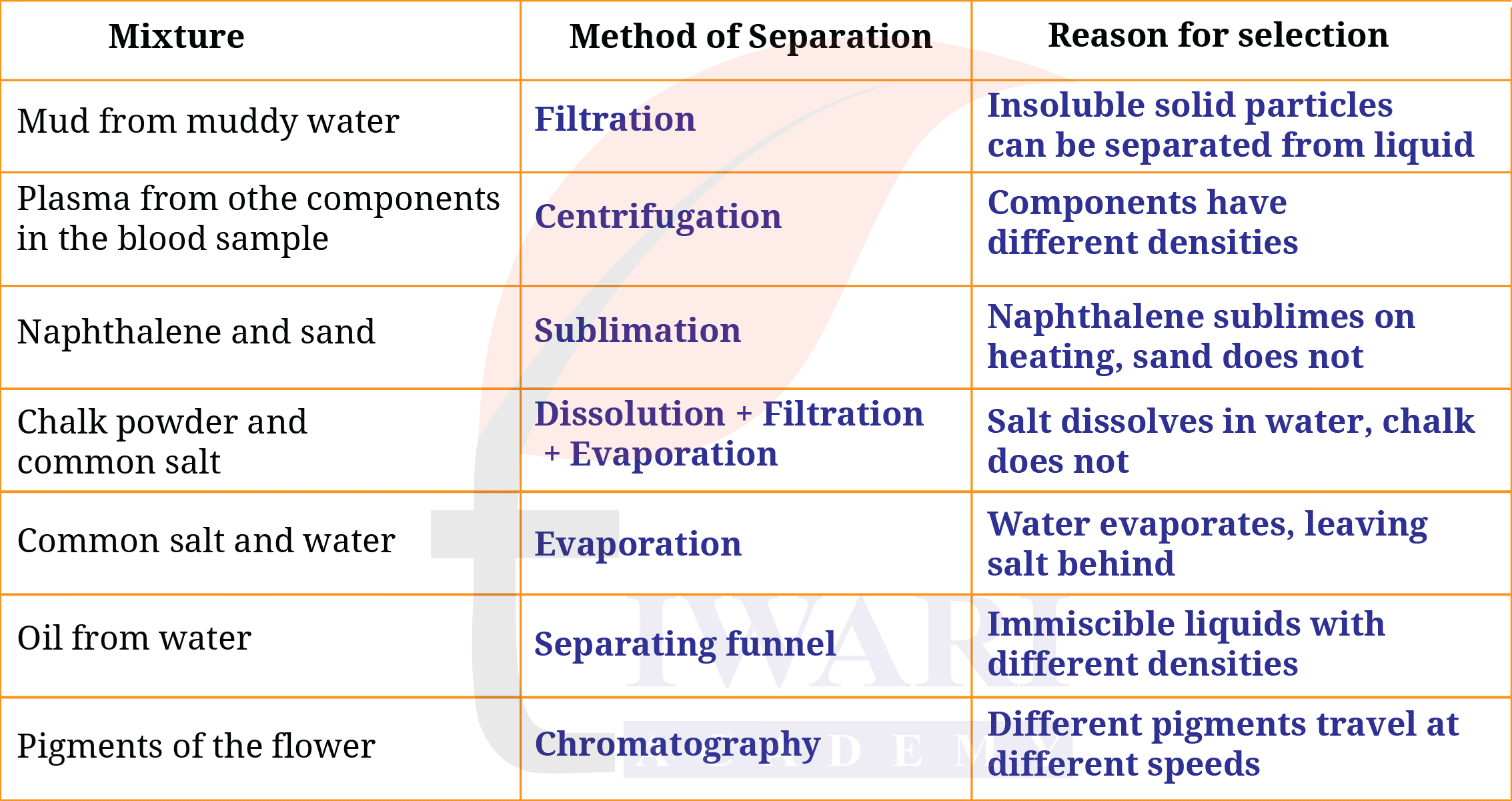
8. Two miscible liquids, A and B, are present in a mixture. The boiling point of A is 60°C and the boiling point of B is 90°C. Suggest a method to separate them. Also, draw a labelled diagram of the method suggested.
Answer:
Method: Simple Distillation
Explanation:
– The difference in boiling points (30°C) is sufficient for separation.
– Liquid A (lower boiling point = 60°C) will vaporise first.
– The vapour is cooled and condensed to collect pure A.
– Liquid B (higher boiling point = 90°C) remains in the flask.

9. Compare evaporation, crystallization and distillation. In which situation would you prefer each of these over the others?
Answer:
Evaporation:
- Used to separate a dissolved solid from a liquid.
- Solvent is lost.
- Example: Obtaining salt from seawater.
- Preferred when purity is not the main concern.
Crystallization:
- Used to obtain pure solid crystals from a solution.
- Removes impurities.
- Example: Copper sulphate crystals.
- Preferred when high purity is required.
Distillation:
- Used to separate liquids or recover solvent.
- Based on difference in boiling points.
- Example: Separation of water and acetone.
- Preferred when both solute and solvent are needed or for liquid-liquid separation.
10. Blood is an example of a colloidal mixture. (i) What would happen if blood behaved like a true suspension inside the body? (ii) In a blood sample, identify the dispersed phase and the dispersion medium.
Answer:
(i) If blood behaved like a true suspension:
- Particles would settle down when left undisturbed.
- Blood cells would separate from plasma.
- This would disrupt circulation and could be life-threatening.
(ii) Identification of the dispersed phase and the dispersion medium:
- Dispersed phase: Blood cells (RBCs, WBCs, platelets)
- Dispersion medium: Plasma
11. You are given a mixture of sand, common salt and naphthalene. The figure depicts various steps used to separate the components of this mixture. Identify and write down the correct sequence of separation techniques.

Answer:
Correct sequence of separation techniques:
1: Sublimation (to separate naphthalene)
3: Dissolution in water (to dissolve salt to separate sand)
2: Evaporation (to obtain salt from solution)
Explanation:
- Naphthalene sublimes on heating, leaving sand and salt behind.
- Salt dissolves in water, sand does not.
- Sand is removed by filtration.
- Salt is obtained by evaporation.
12. Why is distillation an effective method for separating a mixture of water and acetone?
Answer:
- Distillation is effective because water and acetone have different boiling points.
- Acetone boils at about 56°C, while water boils at 100°C.
- The liquid with lower boiling point (acetone) vaporises first and is condensed separately.
- Thus, both liquids can be separated based on difference in boiling points.
13. Answer the following questions with the help of the data given in Table 5.4.
Table 5.4: Solubility of various salts (in g per 100 g of water) at different temperatures
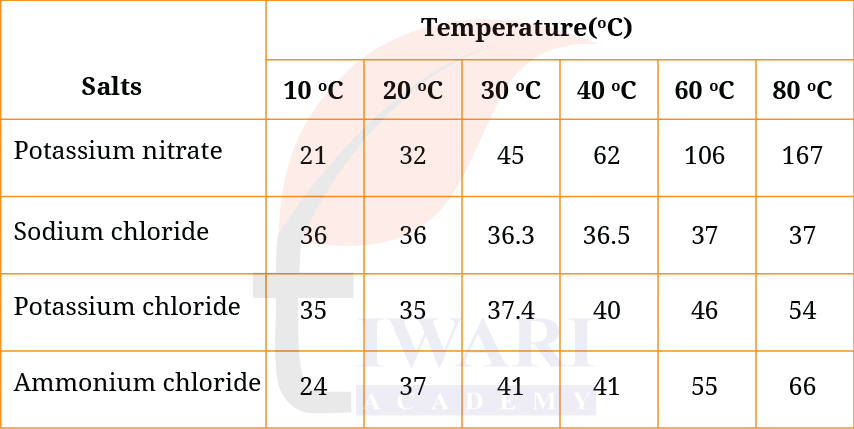
(i) What mass of potassium nitrate would be needed to prepare its saturated solution in 50 g of water at 40°C?
Answer:
From table: At 40°C, solubility of potassium nitrate = 62 g per 100 g water
For 50 g water:
= (62/100) × 50 = 31 g
31 g of potassium nitrate
(ii) A student makes a saturated solution of potassium chloride in water at 80°C and leaves the solution to cool at room temperature (25°C). What would she observe as the solution cools? Explain.
Answer:
- Solubility of potassium chloride decreases on cooling.
- Excess solute comes out of solution.
- Crystals of potassium chloride will form.
(iii) What is the effect of a change in temperature on the solubility of salts? Also, compare the changes in the solubility of the four given salts with increasing temperature from 10°C to 80°C.
Answer:
Effect:
– Generally, solubility of solids increases with increase in temperature.
Comparison:
- Potassium nitrate: Solubility increases sharply (21 → 167 g)
- Sodium chloride: Very little change (36 → 37 g)
- Potassium chloride: Moderate increase (35 → 54 g)
- Ammonium chloride: Considerable increase (24 → 66 g)
14. Three students, A, B and C, are preparing sugar solutions for an experiment:
- Student A dissolves 20 g of sugar in 80 g of water.
- Student B dissolves 20 g of sugar in 100 g of water.
- Student C dissolves 30 g of sugar in 80 g of water.
(i) Calculate the mass percentage (% m/m) concentration of sugar in each student’s solution.
(ii) Whose solution is the most concentrated? Explain why.
Answer:
(i). Formula:
Mass % = (Mass of solute / Mass of solution) × 100
Student A:
Total mass = 20 + 80 = 100 g
Mass % = (20/100) × 100 = 20%
Student B:
Total mass = 20 + 100 = 120 g
Mass % = (20/120) × 100 = 16.67%
Student C:
Total mass = 30 + 80 = 110 g
Mass % = (30/110) × 100 ≈ 27.27%
Final Result:
A = 20%
B = 16.67%
C ≈ 27.27%
(ii). Student C’s solution is the most concentrated because it has the highest mass percentage of sugar (≈ 27.27%).
Explanation:
Higher mass percentage means more solute is present in a given amount of solution, making it more concentrated.
15. Examine Fig. 5.26. (i) Identify the separation technique marked as ‘S’. (ii) Label the apparatus A, B and C. (iii) Which of the following mixtures can be separated by the technique identified above? Use the data given in Table 5.5.
Mixtures:
(a) water — acetone
(b) water — salt
(c) acetone — alcohol
(d) sand — salt
(e) alcohol — chloroform
(f) alcohol — benzene

Answer:
(i) The separation technique marked as ‘S’ is Distillation.
(ii) A → Distillation flask (round-bottom flask)
B → Condenser
C → Receiver flask (conical flask)
Mixtures:
(a) water — acetone
(b) water — salt
(c) acetone — alcohol
(d) sand — salt
(e) alcohol — chloroform
(f) alcohol — benzene
(iii) Mixtures that can be separated by distillation:
(a) Water — Acetone
(e) Alcohol — Chloroform
Explanation:
Distillation is used to separate miscible liquids with different boiling points.
From Table 5.5:
• Water = 100°C, Acetone = 56°C → large difference
• Alcohol = 78°C, Chloroform = 61°C → sufficient difference
Mixtures that cannot be separated by this method:
(b) Water — Salt (solid-liquid mixture → use evaporation)
(c) Acetone — Alcohol (boiling points too close → need fractional distillation)
(d) Sand — Salt (solid-solid mixture → use dissolution + filtration)
(f) Alcohol — Benzene (boiling points close → fractional distillation needed)
Question Bank with Answers | NCERT Session 2026–27
NCERT Class 9 Science Exploration Chapter 5 Very Short Answer Type Questions with Explanation.
Very Short Answer Type Questions
- What is a homogeneous mixture? Give two examples.
Answer:
A homogeneous mixture has a uniform composition throughout. Examples include sugar solution (sugar in water), vinegar (acetic acid in water) and aerated drinks like soda (carbon dioxide in water). - What is the concentration of a solution?
Answer:
Concentration is the amount of solute dissolved in a given amount of solvent or solution. It tells us how much solute is present per unit quantity of the solution or solvent. - Write the formula for mass by mass percentage concentration.
Answer:
Mass by mass percentage (% m/m) = (Mass of solute ÷ Mass of solution) × 100. It tells how many grams of solute are present in 100 grams of the total solution. - What is a saturated solution?
Answer:
A saturated solution is one that cannot dissolve any more solute at a given temperature. The maximum amount of solute that can dissolve at that temperature has already been dissolved. - What is crystallization?
Answer:
Crystallization is the process of forming pure crystals from a saturated solution. It is based on the difference in solubility of a substance at different temperatures and is used to purify solids. - What is distillation? When is it used?
Answer:
Distillation is the process of separating two miscible liquids by heating until the lower-boiling liquid vaporises, then cooling the vapour back to liquid. It is used when liquids differ in boiling point by at least 25°C. - What is the Tyndall effect?
Answer:
The Tyndall effect is the scattering of a beam of light by particles in a colloid or suspension, making the path of light visible. It does not occur in a true solution. It was first explained by scientist John Tyndall. - What is sublimation? Name two substances that sublime.
Answer:
Sublimation is the direct conversion of a solid into vapour without passing through the liquid state. Camphor and naphthalene are common examples. Solid carbon dioxide (dry ice) also undergoes sublimation. - What is the difference between miscible and immiscible liquids? Give one example of each.
Answer:
Miscible liquids mix completely with each other — for example, acetone and water. Immiscible liquids do not mix and form separate layers — for example, mustard oil and water. - What is centrifugation? Where is it commonly used?
Answer:
Centrifugation is the process of spinning a mixture at high speed so that heavier particles settle at the bottom while lighter liquid stays at the top. It is commonly used to separate blood components like RBCs and plasma. - What is coagulation? Name a common coagulant used in water purification.
Answer:
Coagulation is the process by which fine suspended particles are made to clump together by adding a coagulant. Alum (fitkari) is a commonly used coagulant in water purification. - How does the solubility of a solid solute change with temperature?
Answer:
The solubility of a solid solute in a liquid solvent generally increases with increasing temperature. However, the solubility of gases dissolved in liquids generally decreases with increasing temperature. - What is a colloid? Give three examples.
Answer:
A colloid is a mixture in which particles of intermediate size (1–1000 nm) are uniformly dispersed in a medium and do not settle over time. Examples are milk, blood, and tomato sauce. - What is an alloy? Why cannot physical methods separate an alloy?
Answer:
An alloy is a homogeneous mixture of two or more metals, or a metal and a non-metal, formed by melting them together. Physical methods cannot separate alloys because the components are uniformly mixed at the atomic level. - What is paper chromatography? On what principle does it work?
Answer:
Paper chromatography is a technique that separates components of a mixture based on differences in how fast they move through paper with a solvent. Components with stronger interaction with paper move slower and separate from faster-moving ones.
NCERT Class 9 Science Exploration Chapter 5 Short Answer Type Questions with Explanation.
Short Answer Type Questions
- If 15 g of glucose is dissolved in water to make 300 mL of solution, calculate its mass by volume percentage.
Answer:
Mass of glucose (solute) = 15 g
Volume of solution = 300 mL
% m/v = (Mass of solute ÷ Volume of solution) × 100
% m/v = (15 g ÷ 300 mL) × 100 = 5% m/v
This means 5 g of glucose is present in every 100 mL of the solution. - Why is distillation preferred over simple evaporation when we want to recover both the solvent and the solute from a solution?
Answer:
In simple evaporation, the solvent is lost as vapour into the atmosphere and only the solute is recovered. In distillation, the vapour of the lower-boiling liquid is passed through a condenser, cooled back into a pure liquid, and collected separately. This allows recovery of both the solvent and the solute from the mixture, making distillation the preferred method when both components are needed. - Explain how a separating funnel is used to separate mustard oil from water. Why does mustard oil form the upper layer?
Answer:
A mixture of mustard oil and water is poured into the separating funnel and left undisturbed. Two distinct layers form — mustard oil on top and water at the bottom — because mustard oil has a lower density than water. The stopcock is opened slowly to drain out the lower water layer into a container first. The interface portion is discarded, and then the oil layer is collected separately by opening the stopcock again. - What is the difference between sublimation and evaporation? Is sublimation useful in separating mixtures?
Answer:
In evaporation, a liquid converts into vapour. In sublimation, a solid converts directly into vapour without passing through the liquid state. The reverse of sublimation — vapour converting directly back to solid — is called deposition. Sublimation is useful in separating mixtures where one component sublimes and the other does not. For example, camphor can be separated from sand because camphor sublimes on heating while sand remains unchanged. - How does coagulation work in water purification? What role does alum play?
Answer:
Muddy water often contains very fine suspended particles that cannot be removed by simple filtration. When powdered alum (fitkari) is added, it acts as a coagulant — it causes the fine particles to attract each other and clump together into larger aggregates. These larger clumps are heavy enough to settle by gravity through sedimentation. The clear water above can then be separated by decantation or filtered to remove any remaining residue. - A student prepares a saturated solution of a salt at 80°C and then cools it slowly to 20°C. What will the student observe? Name the process and explain its principle.
Answer:
As the solution cools, its capacity to hold the solute decreases because solubility decreases with falling temperature. The excess solute that can no longer remain dissolved separates out as pure solid crystals — this is called crystallization. The principle is the difference in solubility of a substance at different temperatures. The slower the cooling, the larger and more well-shaped the crystals that form. - What is fractional distillation? How is it used in petroleum refining?
Answer:
Fractional distillation is the separation of components of a mixture with relatively small differences (less than 25°C) in their boiling points. In petroleum refining, crude oil is heated in a furnace and the vapours rise through a fractional distillation column. Components with lower boiling points rise higher and condense at cooler levels, while heavier fractions condense lower. This separates crude petroleum into petroleum gas, petrol, kerosene, diesel, lubricating oil, and bitumen. - Why is milk classified as a colloid and not a solution or a suspension? What would happen if blood behaved like a true suspension?
Answer:
Milk is a colloid because its fat droplets have particle sizes in the range of 1–1000 nm — too small to be visible but large enough to scatter light (Tyndall effect) and remain uniformly dispersed without settling. It is not a solution (particles too large) or a suspension (particles do not settle). If blood behaved like a true suspension, the blood cells would settle to the bottom of blood vessels and could no longer be transported efficiently throughout the body, which would be fatal. - Compare evaporation and crystallization. In which situation would you prefer crystallization over simple evaporation?
Answer:
| Feature | Evaporation | Crystallization |
|---|---|---|
| What is recovered | Solute (impure) | Pure solute crystals |
| Solvent recovery | No | No (unless combined with distillation) |
| Temperature | Heated till solvent evaporates | Cooled slowly after saturation |
| Purity of product | Lower | Higher |
| Best used when | Solute is not heat-sensitive | Pure solid crystals are needed |
Crystallization is preferred over evaporation when a pure solid is required, especially when the mixture contains two solids soluble in the same solvent — for example, separating pure copper sulfate from an impure sample.
NCERT Class 9 Science Exploration Chapter 5 Long Answer Type Questions with Explanation.
Long Answer Type Questions
1. Classify mixtures as homogeneous and heterogeneous. Further classify them as solutions, suspensions and colloids. Give examples of each and describe how the Tyndall effect helps distinguish them.
Answer:
Classification of Mixtures:
All mixtures are first classified based on uniformity of composition.
- Homogeneous Mixtures have uniform composition throughout — every part looks and behaves the same. Examples include salt solution, sugar solution, vinegar, aerated drinks and brass alloy.
- Heterogeneous Mixtures are non-uniform — different parts have different compositions. Examples include sand and water, oil and water, smoke and muddy water.
Further Classification — Solution, Suspension, and Colloid:
Solution:
- Particle size less than 1 nm
- Particles not visible to naked eye
- Does not settle on standing
- No Tyndall effect
- Examples: salt in water, copper sulfate solution
Suspension:
- Particle size more than 1000 nm
- Particles visible to naked eye
- Settles on standing undisturbed
- Tyndall effect present
- Examples: sand in water, chalk in water
Colloid:
- Particle size between 1–1000 nm
- Particles not visible to naked eye
- Does not settle on standing
- Tyndall effect present
- Examples: milk, blood, fog, starch in water
Comparison Table:
| Property | Solution | Colloid |
|---|---|---|
| Particle size | Less than 1 nm | 1–1000 nm |
| Settling | Does not settle | Does not settle |
| Tyndall effect | Absent | Present |
| Property | Suspension | Colloid |
|---|---|---|
| Particle size | More than 1000 nm | 1–1000 nm |
| Visibility | Visible to naked eye | Not visible |
| Settling | Settles on standing | Does not settle |
The Tyndall Effect as a Distinguishing Test:
When a beam of laser light is passed through the three types of mixtures, the behaviour of light clearly distinguishes them. In a solution, the beam passes straight through without any visible path because particles are too small to scatter light. In a suspension, the beam path is clearly visible because large particles scatter light in all directions. In a colloid, the beam is also visible as intermediate-sized particles scatter light — this is the Tyndall effect.
Real-life examples of the Tyndall effect include sunlight entering a dark room through a small opening scattered by dust particles, fog lights appearing as bright cones and floodlights in a sports stadium appearing as visible beams.
2. Describe the three methods of expressing concentration of a solution. Give one example and one real-life application of each method.
Answer:
Concentration is the amount of solute dissolved in a given amount of solvent or solution. Here, three percentage-based methods are described.
A. Mass by Mass Percentage (% m/m)
Formula: % m/m = (Mass of solute ÷ Mass of solution) × 100
Example: If 10 g of salt is dissolved in 90 g of water:
Mass of solution = 10 + 90 = 100 g
% m/m = (10 ÷ 100) × 100 = 10% m/m
Real-life application: Used on food product labels — milk powder packets show fat, sugar and protein content as % m/m per 100 g of product.
B. Mass by Volume Percentage (% m/v)
Formula: % m/v = (Mass of solute ÷ Volume of solution) × 100
Example: If 5 g of glucose is dissolved to make 100 mL of solution:
% m/v = (5 g ÷ 100 mL) × 100 = 5% m/v
Real-life application: Used in medicines and hospital drips — glucose IV solution is labelled 5% w/v and saline drips are 0.9% m/v sodium chloride in water.
C. Volume by Volume Percentage (% v/v)
Formula: % v/v = (Volume of solute ÷ Volume of solution) × 100
Example: If 1 mL of pesticide is dissolved to make 100 mL of spray solution:
% v/v = (1 mL ÷ 100 mL) × 100 = 1% v/v
Real-life application: Used for liquid-liquid mixtures — vinegar is labelled 5% v/v acetic acid and perfumes express concentration of fragrant oils in % v/v.
Choosing the Right Method:
| Method | Best Used When | Example |
|---|---|---|
| % m/m | Both solute and solvent measured by mass | Food labels, milk powder |
| % m/v | Solute by mass, solution by volume | Medicines, IV drips |
| % v/v | Both solute and solution are liquids | Vinegar, perfumes |
3. Describe the process of crystallization in detail. Explain the principle, the steps involved in preparing copper sulfate crystals and its applications in daily life and industry.
Answer:
Crystallization: Crystallization is the process of forming a pure solid substance in the form of crystals from its saturated solution by cooling it slowly. A crystal is a solid in which particles are arranged in a regular geometric pattern.
Principle:
The principle of crystallization is based on the difference in solubility of a substance at different temperatures. The solubility of most solid solutes increases with temperature. When a hot saturated solution is cooled, the excess solute that can no longer remain dissolved separates out as pure crystals. Slower cooling produces larger and better-shaped crystals than rapid cooling.
Steps for Preparing Copper Sulfate Crystals:
- Dissolve 1 g of copper sulfate in 25 mL of water with a drop of dilute sulfuric acid to prevent impurities.
- Heat gently in a water bath, stirring constantly and add more copper sulfate until the solution becomes saturated.
- Filter the hot saturated solution to remove insoluble impurities.
- Collect the filtrate in a clean beaker and cover with a watch glass.
- Allow the solution to cool slowly without disturbing it.
- Blue, shiny, well-shaped copper sulfate crystals form gradually.
- Filter the crystals, rinse with cold water and dry on a watch glass.
Comparison of Slow and Rapid Cooling:
| Condition | Crystal Size | Crystal Shape |
|---|---|---|
| Slow cooling at room temperature | Larger | Well-formed and regular |
| Rapid cooling in ice-cold water | Smaller | Poorly formed and irregular |
Applications of Crystallization:
- Salt production from seawater – seawater is evaporated to form a concentrated solution, which is then crystallized to obtain salt.
- Purification of impure solids – crystallization deposits only the pure desired compound, leaving impurities behind in solution.
- Preparation of mishri (candy sugar) – sugar syrup is concentrated and crystallized slowly to form large sugar crystals.
- Ancient Indian salt making – coastal communities produced panga salt by boiling concentrated sea brines and karkatch salt by evaporating seawater, both based on crystallization principles.
- Natural crystal formation – snowflakes, frost on windows and rock salt deposits are all formed by natural crystallization.
Frequently Asked Questions – FAQs
Is Class 9 Science Exploration Chapter 5 from the new NCERT book? How is it different from the old syllabus?
Yes, Class 9 Science Exploration Chapter 5 — Exploring Mixtures and their Separation — is from the new NCERT Exploration textbook for Session 2026–27. The older NCERT Science book had a chapter called “Is Matter Around Us Pure?” which covered similar topics but in a more theoretical way.
The new Exploring Mixtures and their Separation chapter adds several things not in the older book — three methods of expressing concentration with formulas and examples, paper chromatography as a hands-on activity, the paperfuge and its medical application, India’s scientific contributions on salt crystallization and the Deg-Bhapka distillation method from Kannauj and a detailed section on colloids, emulsions and the Tyndall effect. Students following the 2026–27 session must use the new Exploration textbook.
Is Class 9 Science Exploration Chapter 5 easy or difficult?
Class 9 Science Exploration Chapter 5 is considered moderately easy by most students. The concepts — solutions, suspensions, colloids and separation techniques — connect directly to everyday life like making tea, filtering water, and cooking.
The slightly harder parts are the three concentration formulas (% m/m, % m/v, % v/v) and remembering which separation technique applies to which type of mixture. Students who enjoy practical science will find Exploring Mixtures and their Separation one of the more interesting chapters in Class 9, because almost every concept has a kitchen, medical, or industrial example attached to it.
What are the main difficulties students face in Class 9 Science Exploration Chapter 5?
The three main difficulties in Exploring Mixtures and their Separation are predictable and fixable.
- First, students confuse the three concentration formulas — mass by mass, mass by volume and volume by volume — and apply the wrong one to numerical problems. The key is to read the question carefully and check what units are given.
- Second, students mix up solution, suspension and colloid, especially when it comes to the Tyndall effect and particle sizes.
- Third, students struggle to decide which separation technique to use for a given mixture — this becomes easy once the property being exploited for each technique is clearly understood rather than memorised.
How many formulas does my child need to learn in Class 9 Science Exploration Chapter 5?
There are exactly three concentration formulas in Exploring Mixtures and their Separation. The first is mass by mass percentage, which equals mass of solute divided by mass of solution multiplied by 100. The second is mass by volume percentage, which equals mass of solute divided by volume of solution multiplied by 100.
The third is volume by volume percentage, which equals volume of solute divided by volume of solution multiplied by 100. These three formulas, along with a clear understanding of what solute and solvent mean, are the only calculations required. The rest of Class 9 Science Exploration Chapter 5 is conceptual and does not involve heavy mathematics.
How is Class 9 Science Exploration Chapter 5 different from the old NCERT “Is Matter Around Us Pure?” chapter?
The differences are substantial in both content and approach. The old chapter covered solutions, suspensions, colloids and basic separation methods in a mostly descriptive format. The new Exploring Mixtures and their Separation chapter introduces three quantitative methods of expressing concentration — % m/m, % m/v, and % v/v — with worked examples, which was not covered at this level before.
New separation techniques added include paper chromatography with a hands-on activity, centrifugation and coagulation for heterogeneous mixtures and sublimation with the camphor-sand experiment. The chapter also adds a Bridging Science and Society section on the paperfuge for malaria detection, India’s Scientific Contributions on Kannauj’s Deg-Bhapka distillation and ancient salt crystallization, and a detailed section on emulsions and emulsifying agents. Teachers familiar with the old curriculum should plan extra periods for the concentration numericals and the new separation techniques sections.
The concentration numericals in Class 9 Science Exploration Chapter 5 are new to this curriculum. How should teachers introduce them effectively?
The most effective approach is to start with real-life labelled products before introducing the formulas. Bring in a bottle of glucose intravenous solution labelled 5% w/v, a packet of milk powder with % m/m nutrition information and a bottle of vinegar labelled 5% v/v acetic acid — these are the exact examples used in the chapter.
Ask students to read the labels and discuss what the percentages mean before any formula is introduced. This makes the three different methods feel logical rather than arbitrary. When students understand that % m/m is used for solid-solid or solid-liquid by weight, % m/v is used when liquid volume is easier to measure, and % v/v is used for liquid-liquid mixtures, choosing the right formula in problems becomes a matter of reasoning rather than memorisation.
Is Class 9 Science Exploration Chapter 5 easy to score in exams?
Yes, Exploring Mixtures and their Separation is one of the more scoring chapters in Class 9 Science if prepared correctly. The separation techniques section is almost entirely application-based and follows clear rules — once you know which property each technique exploits, exam questions become predictable.
The concentration numericals follow three fixed formulas and are straightforward once practised. The Tyndall effect, colloids and properties of solutions, suspensions and colloids are favourite comparison-table questions in exams. Students who understand the logic behind each separation method, rather than memorising the names, consistently score well in Class 9 Science Exploration Chapter 5.
How can I complete Class 9 Science Exploration Chapter 5 in 2 days?
Completing Exploring Mixtures and their Separation in 2 days is very achievable with a clear plan.
On Day 1, study sections 5.1 to 5.3 — classification of mixtures, solutions, concentration formulas with all three worked examples, solubility curves, crystallization, distillation and paper chromatography. Solve all the Pause and Ponder questions and the numerical examples in these sections.
On Day 2, cover sections 5.4 and 5.5 — separation of heterogeneous mixtures including immiscible liquids, sublimation, suspensions, centrifugation, coagulation, colloids and the Tyndall effect. Finish by solving the Revise Reflect Refine questions, especially the table-based and separation-method questions.
Two focused days are sufficient to cover all core content of Class 9 Science Exploration Chapter 5.
Is it possible to complete Class 9 Science Exploration Chapter 5 in one day?
Completing Exploring Mixtures and their Separation in one day is possible for revision but not for first-time learning. The chapter has concentration numericals, multiple separation techniques and three distinct types of mixtures — solutions, suspensions and colloids — each with their own properties and examples. Trying to learn all of this for the first time in one day will result in shallow understanding and poor performance in application questions.
However, if you have already read Class 9 Science Exploration Chapter 5 once and need a quick revision before an exam, one focused day works well — spend the morning on concepts and tables, the afternoon on numericals and the evening on revision of separation techniques and their principles.
How can I solve concentration problems in Class 9 Science Exploration Chapter 5 easily?
Solving concentration numericals in Exploring Mixtures and their Separation becomes easy with one simple habit — identify what is given before picking a formula. If the problem gives you mass of solute and mass of solution or solvent, use % m/m. If it gives mass of solute and volume of solution in mL, use % m/v. If it gives volume of solute and volume of solution, use % v/v.
Always remember that in % m/m, the denominator is the total mass of solution, not just the solvent — so if 10 g of salt is dissolved in 90 g of water, the mass of solution is 100 g, not 90 g. This single mistake accounts for most wrong answers in Class 9 Science Exploration Chapter 5 concentration problems.
What is the Tyndall effect and why does it keep coming in exams from Class 9 Science Exploration Chapter 5?
The Tyndall effect is the scattering of a beam of light by the particles in a colloid or suspension, making the path of light visible. It does not occur in a true solution because the particles are too small to scatter light. Named after scientist John Tyndall, this effect is observable in everyday life — sunlight through leaves, floodlights in a stadium, and headlights in fog all show the Tyndall effect.
It comes repeatedly in exams from Exploring Mixtures and their Separation because it is the single most reliable test to distinguish a colloid from a solution. Any question asking you to identify a colloid, or explain why milk looks different from salt water in a laser beam, is testing your understanding of the Tyndall effect.


